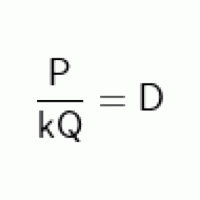tallest_goblin
New member
- Joined
- May 26, 2024
- Messages
- 2
Hello - I am currently studying to become a water treatment operator via correspondence course and as part of the program, I am working to relearn basic algebra. The job requires a lot of unit conversion and dosage calculation, and I haven't had practical experience with mathematics since my high school days.
One of my sample problems includes rearranging the standard formula for calculating a chemical dosage via unit conversions. That formula looks like this:
P [lb/d] = Q [Mgal/day] x D [mg/L] x 3.785L/gal x 10^6/M x kg/10^6mg x 2.205lb/kg
Where P is the total mass of chemical used in a day, Q is the total volume of water treated per day in millions of gallons (Mgal), and D is the desired dosage of chemical in milligrams per Liter of water.
I have gotten used to this conversion formula, but I am having trouble rearranging it to find different variables (the textbook, frankly, does almost nothing to explain these formulae). In the sample problem, an operator is rearranging the formula to solve for the dosage [D] based on the total chemical used in a day [P]. The rearranged formula has D moved to the left of the equals sign, and on the right, P is in the numerator being divided by the entirety of the remaining formula.
My question is this: my understanding is that to rearrange the equation, I would need to 1) divide both sides by D to move it to the left side, then 2) multiply both sides by P, but that wouldn't leave it in the numerator of the right side. I know I'm missing something obvious, but as I said the textbook does nothing to explain these rearrangements, it only displays them. How can I properly conceptualize this formula so that I can solve for other variables within it? Thank you.
One of my sample problems includes rearranging the standard formula for calculating a chemical dosage via unit conversions. That formula looks like this:
P [lb/d] = Q [Mgal/day] x D [mg/L] x 3.785L/gal x 10^6/M x kg/10^6mg x 2.205lb/kg
Where P is the total mass of chemical used in a day, Q is the total volume of water treated per day in millions of gallons (Mgal), and D is the desired dosage of chemical in milligrams per Liter of water.
I have gotten used to this conversion formula, but I am having trouble rearranging it to find different variables (the textbook, frankly, does almost nothing to explain these formulae). In the sample problem, an operator is rearranging the formula to solve for the dosage [D] based on the total chemical used in a day [P]. The rearranged formula has D moved to the left of the equals sign, and on the right, P is in the numerator being divided by the entirety of the remaining formula.
My question is this: my understanding is that to rearrange the equation, I would need to 1) divide both sides by D to move it to the left side, then 2) multiply both sides by P, but that wouldn't leave it in the numerator of the right side. I know I'm missing something obvious, but as I said the textbook does nothing to explain these rearrangements, it only displays them. How can I properly conceptualize this formula so that I can solve for other variables within it? Thank you.